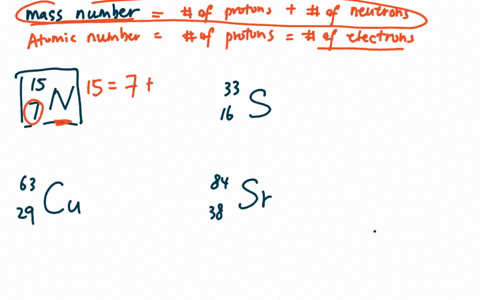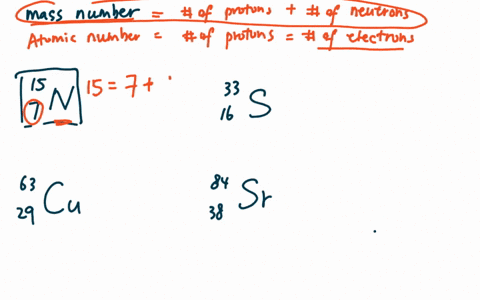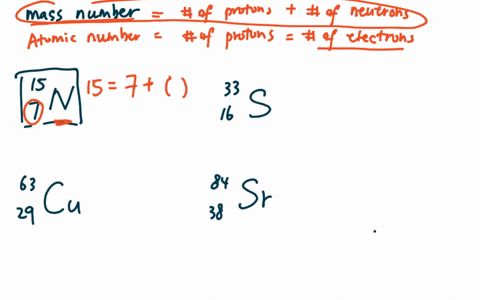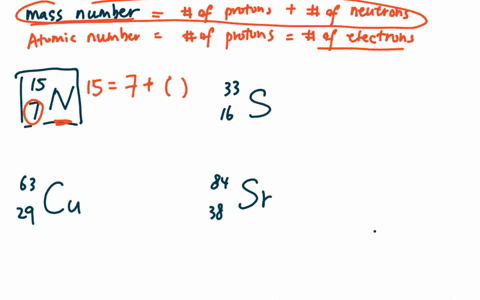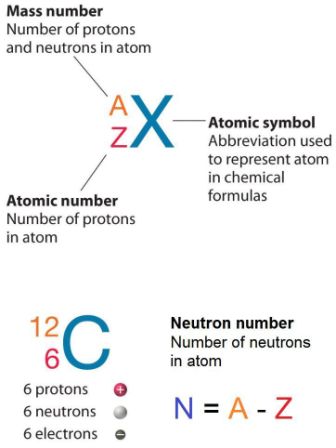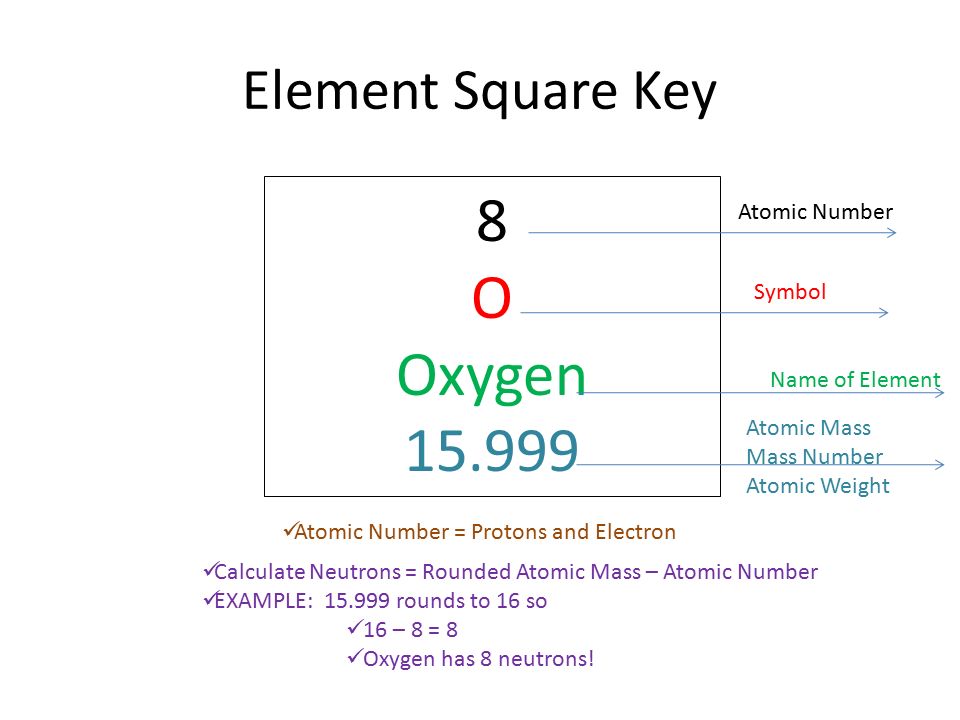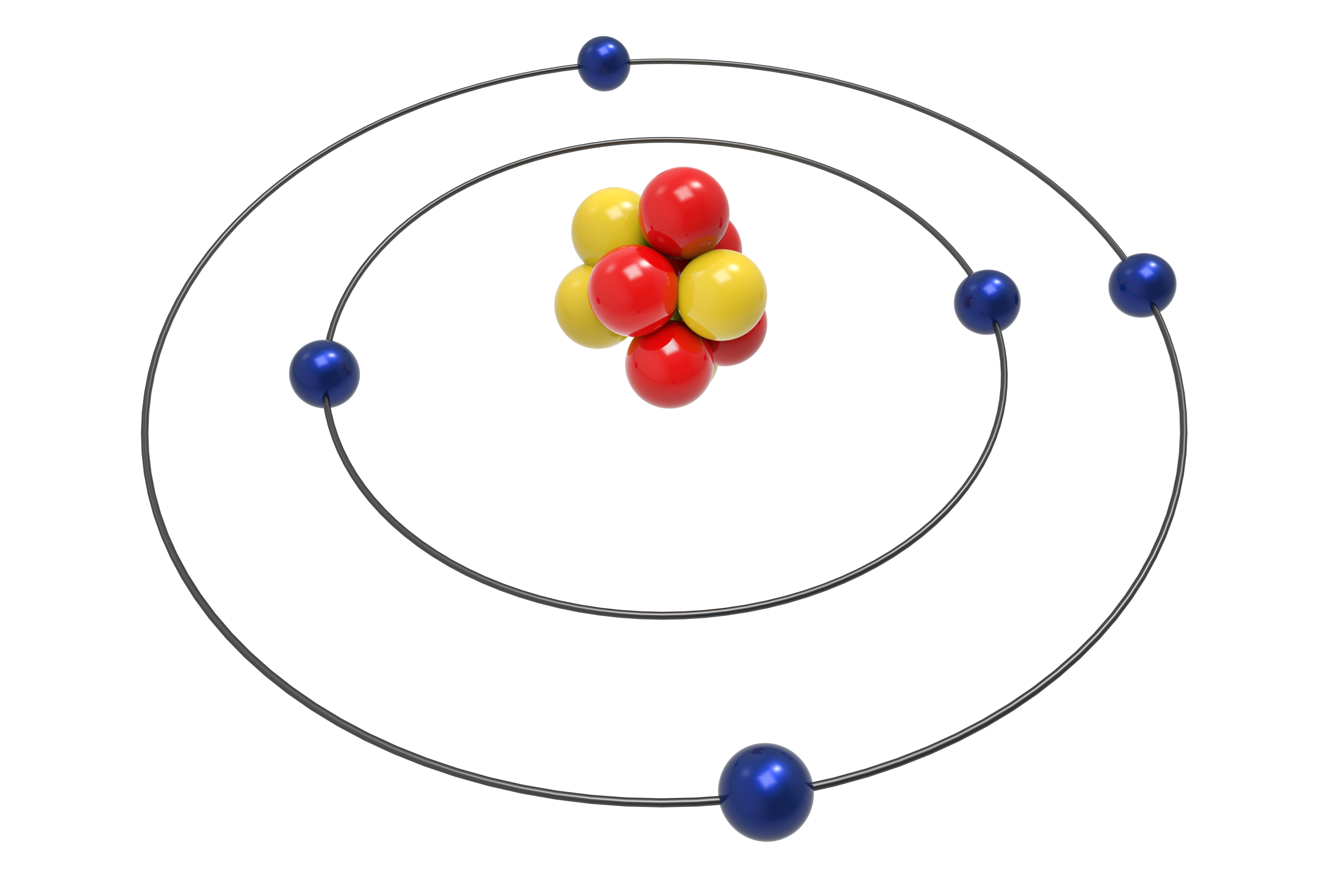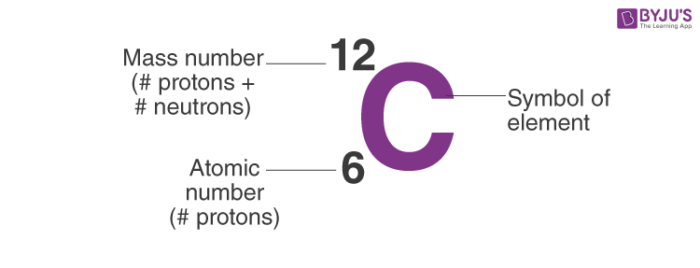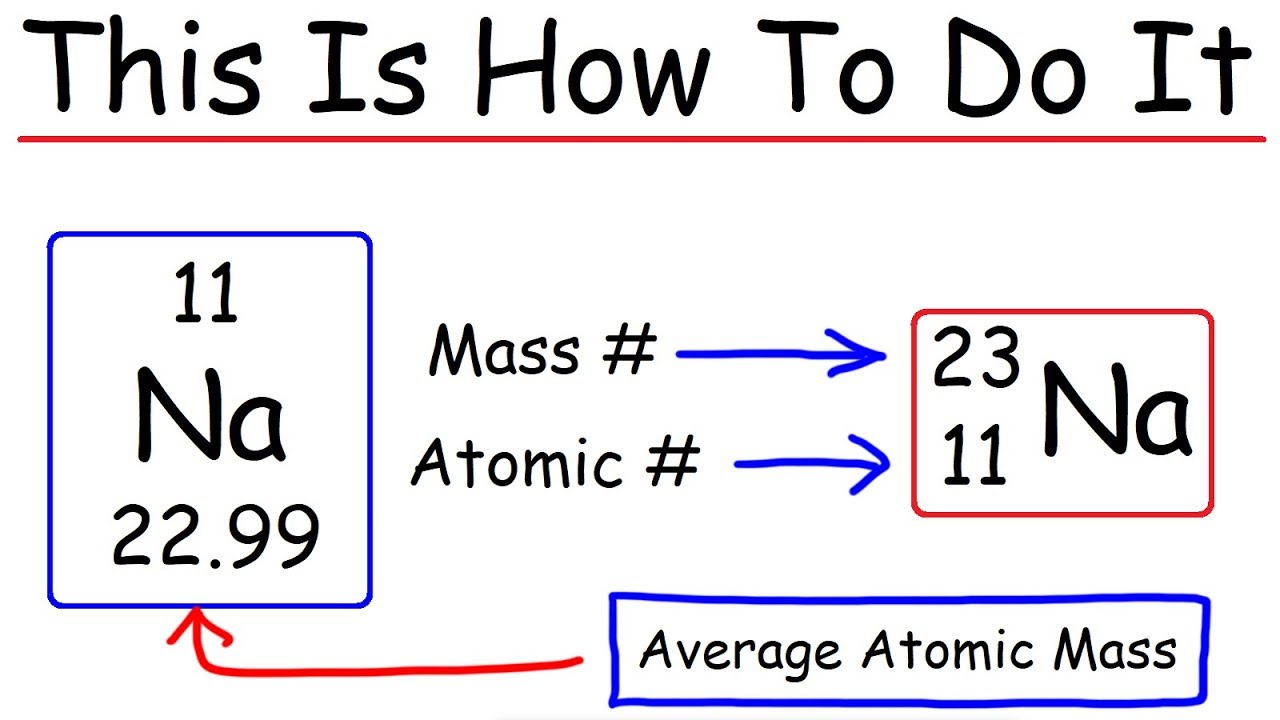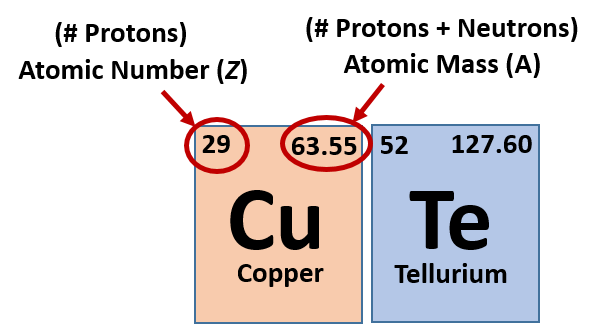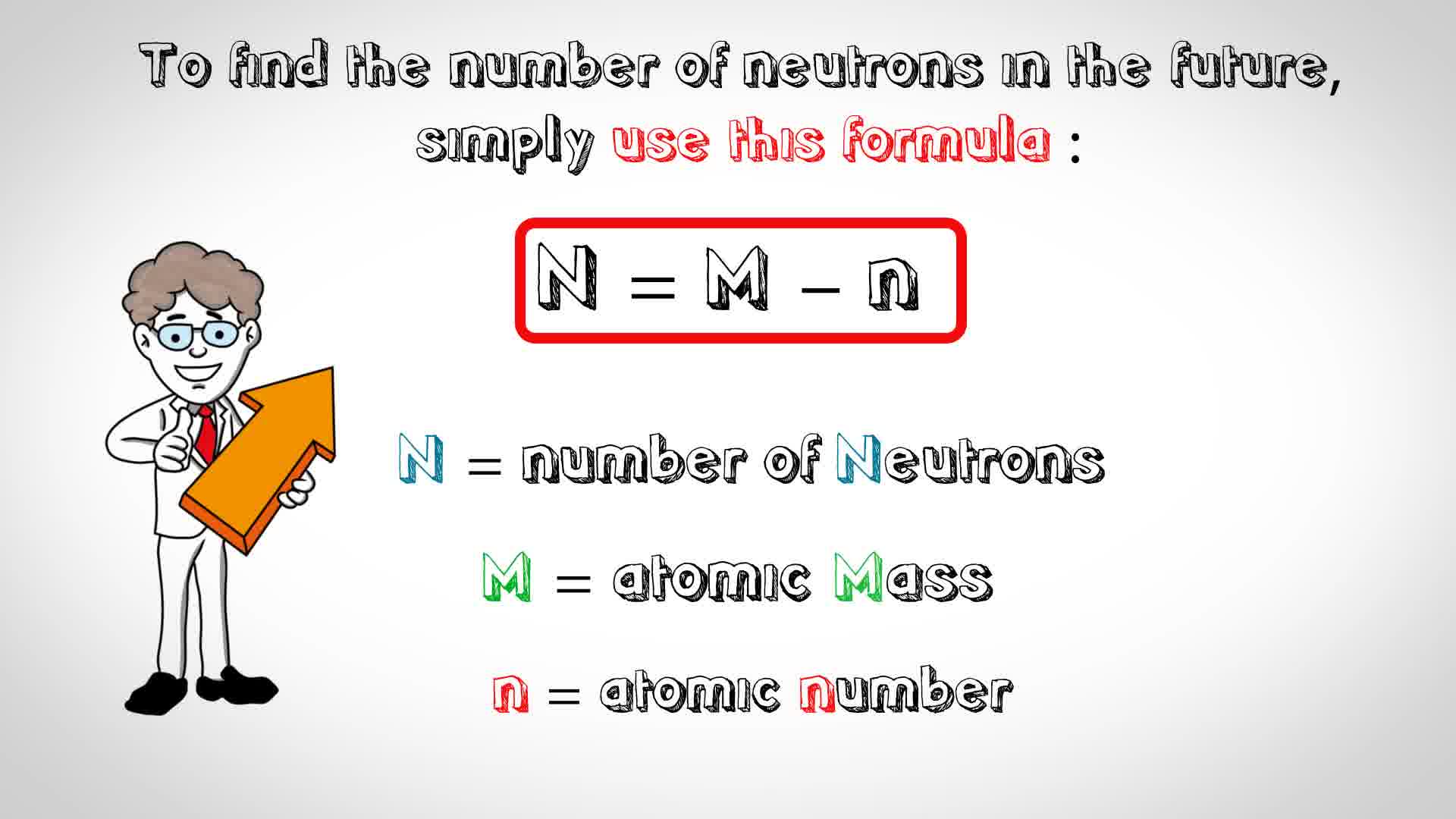
Zirconium has an atomic number of 40. How many neutrons are there in an isotope of zirconium-92? | Socratic
Solved] Calculate the number of protons, neutrons, and electrons in a sample of 209Bi5+ that has the same mass as the bismuth sample you weighed. Th... | Course Hero

Calculate the number of neutrons in the following atoms : (a) ""(27)^(13)AI (b) ""(31)^(15)P ( c) ""(190)^(76)O (d) ""(54)^(24)Cr

Calculate the number of protons, electrons, neutrons in Bromine with atomic number 35 and mass number 80.
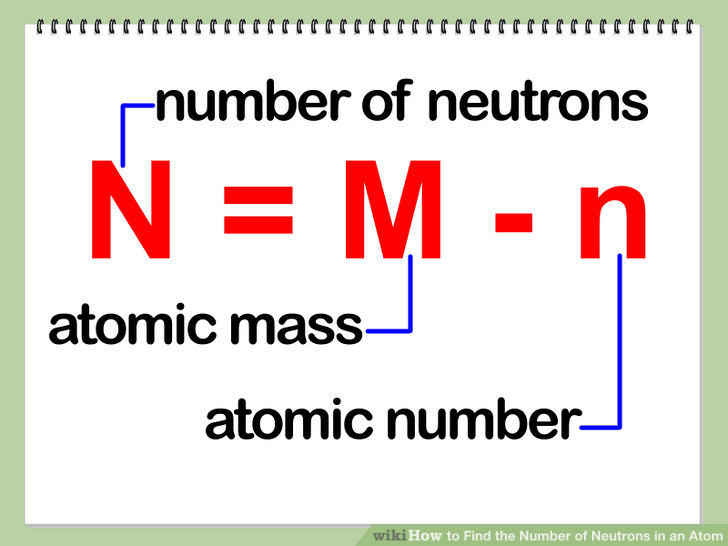
The mass number of a chromium atom is 52 and it has 24 protons. How many neutrons does this atom have? | Socratic